LETTER
BMI and Waist Circumference Have Positive Correlation With Mitochondrial DNA Copy Number in Young Adult Male
Liong Boy Kurniawan1, *, Uleng Bahrun1, Mochammad Hatta2, Mansyur Arif1
Article Information
Identifiers and Pagination:
Year: 2018Volume: 8
First Page: 1
Last Page: 6
Publisher Id: TOOBESJ-8-1
DOI: 10.2174/1876823701808010001
Article History:
Received Date: 28/02/2018Revision Received Date: 19/04/2018
Acceptance Date: 10/05/2018
Electronic publication date: 31/05/2018
Collection year: 2018
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:
Abdominal obesity is associated with metabolic and cardiovascular diseases. Mitochondrial dysfunction is often related to metabolic disorder commonly found in abdominal obesity. Mitochondrial DNA (mtDNA) copy number is a biomarker which can be used to reflect the activity of mitochondria. Abdominal obesity is more commonly found in males compared to females and the incidence is growing in young adult male, therefore we investigated the relationship of mtDNA copy number and anthropometric and metabolic biomarkers in young adult male.
Methods:
A total of 25 healthy young adult males of age 19 to 24 years old were recruited in the study. Metabolic biomarkers were evaluated along with body mass index, waist circumference and total body fat, visceral fat by bioelectrical impedance analysis. mtDNA copy number was measured in peripheral blood leukocytes by using real-time polymerase chain reaction method.
Results:
After adjusting for ages, mtDNA copy number correlated with body mass index (r= 0.45, P= 0.03) and waist circumference (r= 0.43, P= 0.04) but had no significant correlation with fasting plasma glucose, insulin, HOMA-IR, testosterone, total body fat and visceral fat.
Conclusion:
mtDNA copy number has a positive correlation with BMI and waist circumference. Even though it is still controversial over other studies, further studies are needed to explain the causality and significance of our findings.
1. INTRODUCTION
Obesity prevalence is increasing worldwide in developed and developing countries including Indonesia [1, 2]. Obesity is linked to low-grade chronic inflammation which could lead to several health disorders such as cardiovascular disease (CVD), Diabetes Mellitus (DM), arthritis and other diseases [3, 4]. Abdominal obesity which is more commonly found in male than female is a major source of pro-inflammatory cytokines and adipokines. Excess of visceral fat is associated with worse metabolic profile than other body fat deposition [5].
Mitochondria are essential double membrane subcellular organelles with the primary function to support aerobic respiration and produce energy of the cells. Mitochondria are the main source of adenosine triphosphate (ATP) which drives fundamental cell functions such as muscle contraction, cell divisions, etc. Mitochondria play a fundamental role in human health [6-8]. Dysfunction of mitochondria is associated with several diseases related to aging such as insulin resistance, DM, and CVD [9].
Mitochondrial Deoxyribonucleic Acid (mtDNA) copy number reflects mitochondrial DNA; a double stranded, circular molecule of 16,569 bp contains 37 genes. mtDNA encodes polypeptide of all subunits of enzyme complexes of the oxidative phosphorylation system thus reduced mtDNA copy number reflects dysfunction of mitochondria [10]. Reduction in mtDNA copy number in peripheral blood was associated with visceral adiposity [11], diabetic nephropathy [12], chronic kidney disease [13], Parkinson’s disease [14], and prostate cancer [15]. Adipose tissue produces pro-inflammatory cytokines which increase oxidative stress and may disturb mitochondrial function. Some previous studies have reported mixed results of the effect of obesity on mtDNA copy number.
In this study, we investigated the association of peripheral blood mtDNA copy number with the Body Mass Index (BMI), waist circumference, total body fat, visceral fat and other metabolic markers in healthy young adult males with age of 19 to 24 years.
2. MATERIALS AND METHODS
2.1. Study Sample
This was a cross-sectional study performed on voluntary participants during the period of July 2017 to February 2018. The participants were the 1st to the 6th year male medical students of Hasanuddin University, Makassar, Indonesia. Written informed consent was obtained from each participant. We recruited 25 male subjects with age from 19 to 24 years old. We excluded participants who used medication including oral hypoglycemic agents, lipid-reducing drugs and corticosteroids. This study was approved by Komite Etik Penelitian Kesehatan (Health Research Ethical Committee) of Medical Faculty, Hasanuddin University and complied with the Declaration of Helsinki.
2.2. Measurements
Anthropometric measurements were performed by a single examiner. Body mass index was calculated as weight (kg) divided by height squared (m2). Total body fat and visceral fat were measured by using Tanita (Tokyo, Japan) bioelectrical impedance analysis. Blood samples were collected after at least 8 hours overnight fasting period. Fasting glucose was measured using Abx Pentra 400 (Horiba, USA) while insulin and testosterone were measured using Elecsys 2010 (Roche, Indianapolis, IN, USA). Insulin resistance was calculated using the homeostatic model assessment of insulin resistance (HOMA-IR) index= (Insulin [µIU/mL] x Fasting Blood Glucose [mg/dL]/ 405.
2.3. Measurement of Mitochondrial DNA Copy Number in Peripheral Blood
mtDNA copy number extraction from peripheral blood leukocyte was performed according to Boom method [16]. The relative mtDNA copy number was measured by a real-time polymerase chain reaction (qPCR) according to the method of Wong and Cortopassi [17], Liu et al. [18]. Reactions were performed using a light cycler-fast start DNA Master SYBR Green I kit purchased from Roche Applied Science (Pleasanton, CA, USA). The forward and reverse primers of β Globin (Housekeeping gene) to amplify a 268 bp product were 5’-GAAGAGCCAAGGACAGGTAC-3’ and 5’-CAACTTCATCCACGTTCAACC-3’ respectively. The forward and reverse primers of the mitochondrial gene (ND1 gene) used to amplify 153 bp products were 5’-AACATACCCATGGCCAACCT-3’ and 5’-AGCGAAGGGTTGTAGTAGCCC-3’ respectively. After denaturation at 95oC for 300 seconds, DNA samples were treated with 95oC for 0.1 seconds, 58oC for 6 seconds and 72oC for 18 seconds for 40 cycles. A total of 20 ng of DNA was used and the number of PCR cycles to reach this amount of DNA was defined as threshold cycles (ct). The following equation was used to quantify the mtDNA copy number to β Globin relative copy number= 2Δct (Δct= ct β Globin - ct ND1).
2.4. Statistical Analysis
Normality of data distribution is tested by using Shapiro-Wilk test. Normally distributed data are expressed as mean+standard deviation (SD). Non normally distributed data are expressed as median (minimum-maximum). Testosterone levels and total body fat are normally distributed while age, BMI, waist circumference, fasting plasma glucose, insulin, HOMA-IR, visceral fat and mtDNA copy number (ND1) are not normally distributed. Correlation between mtDNA copy number and all variables was analyzed with Spearman Test, followed by then continued by partial correlation after controlling for age. All statistical analyses are performed by using the Statistical Package for the Social Sciences, Version 21.0 (SPSS Inc, Chicago, IL, USA). Statistical significance was defined as p<0.05.
3. RESULTS
The characteristic of study subjects is shown in Table 1. The median (min-max) age of the study subjects was 20 (19-24) years while mtDNA copy number was 9.56 (8.07-12.34). The correlation between mtDNA copy number with other variables is shown in Table 2. mtDNA copy number had no significant correlation with other variables, but after adjusting for age, mtDNA copy number positively correlated with BMI (r= 0.45, P= 0.03) and waist circumference (r= 0.43, P= 0.04) shown in Fig. (1).
| Variables | Total (n=25) |
|---|---|
| Age (Years)* | 20 (19-24) |
| BMI (Kg/m2)* | 24.99 (16.33-42.98) |
| Waist Circumference (cm)* | 92 (67-132) |
| Total Body Fat (%) | 22.03+8.69 |
| Visceral Fat | 8 (1-19) |
| Fasting Glucose (mg/dL)* | 99 (80.20-110.80) |
| Fasting Insulin (µIU/mL)* | 14.29 (5.1-45.1) |
| HOMA-IR* | 3.35 (1.22-11.29) |
| Testosterone (ng/mL) | 5.63+1.41 |
| mtDNA Copy Number* | 9.56 (8.07-12.34) |
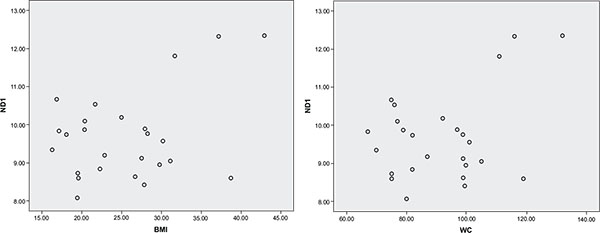 |
Fig. (1). The Relationship Between Body Mass Index (kg/m2), Waist Circumference (cm) and mtDNA Copy Number (ND1). |
| Variables | Unadjusted | Age Adjusted | ||
|---|---|---|---|---|
| r | P-Value | r | P-Value | |
| Age (Years) | -0.10 | 0.64 | - | - |
| BMI (Kg/m2) | 0.13 | 0.52 | 0.45 | 0.03 |
| Waist Circumference (cm) | 0.06 | 0.76 | 0.43 | 0.04 |
| Total Body Fat | -0.38 | 0.86 | 0.28 | 0.19 |
| Visceral Fat | 0.10 | 0.65 | 0.38 | 0.07 |
| Fasting Glucose (mg/dL) | -0.19 | 0.36 | -0.26 | 0.22 |
| Fasting Insulin (µIU/mL) | 0.17 | 0.43 | 0.19 | 0.38 |
| HOMA-IR | 0.18 | 0.40 | 0.13 | 0.53 |
| Testosterone (ng/mL) | -0.13 | 0.54 | -0.32 | 0.13 |
4. DISCUSSION
Mitochondria are essential organelles with the primary function to support aerobic respiration and produce ATP which drive fundamental cell functions [7]. These functions include force generation (muscle contraction, cell division, etc), biosynthesis, degradation of proteins, generation and maintenance of membrane potentials [8]. Mitochondrial dysfunction induces several metabolic disorders such as insulin resistance and CVD [9]. Several diseases have been linked to mitochondrial dysfunction shown by the change of mtDNA copy number [19]. It has been reported that reduced mtDNA copy number is associated with various conditions such as visceral adiposity [11], impaired glucose-stimulated islet β cell function [20], diabetic nephropathy [12], chronic kidney disease [13], Parkinson’s disease [14], and prostate cancer [15]. Instead, various mixed results have been reported. Elevated mtDNA copy number is reported to be associated with childhood autism [21]. mtDNA copy number is also reported to be increased in diabetes patients compared with healthy controls, even the increase is considered non-functional [12].
The association of mtDNA copy number with adipose and obesity showed mixed results. It has been reported that visceral adipose has lower mitochondrial respiration than subcutaneous adipose tissue [22]. Lee at al. reported an independent inverse association with mtDNA content and visceral adiposity. The investigator proposed some possible mechanisms. First, visceral adipose tissue secretes pro-inflammatory cytokine which induces mitochondrial dysfunction. Second, free fatty acids’ accumulation in visceral adipose tissue elevates oxidative stress driving to mitochondrial dysfunction [11].
In contrary, our study showed the opposite result. Initially, we found no correlation between peripheral leukocyte mtDNA copy number with, BMI, waist circumference, total body fat, visceral fat, HOMA-IR and testosterone, but after controlling the age, mtDNA copy number had a positive correlation with BMI and waist circumference but not with total body fat and visceral fat. In line with our result, Naeyer et al. reported that mtDNA copy number was higher in visceral adipose tissue of obese subjects than in control [23]. Based on this mixed result, the association between obesity and mtDNA copy number remains unclear. Even though our result showed a positive correlation between mtDNA copy number and waist circumference, but we could not determine the causality. Our cross-sectional study could not reveal the causal factor of our finding. We proposed some possible mechanisms which might explain our result.
First, the increase in mtDNA copy number might reflect a compensatory mtDNA over replication mechanism in healthy young adult male subjects with narrower age lifespan (18-24 years) compared with the previous study by Lee et al. with wider age span (20-40 years) [11]. We hypothesized that in very young adults, there is still better compensatory mechanism while in older adults, the mtDNA copy number would decrease along with obesity and aging. This theory still must be confirmed by further studies. Another explanation is that the increase in mtDNA copy number might be not fully functional as mtDNA copy number function is to encode mitochondrial RNAs. There might be a disconnection between the increase in mtDNA and mitochondrial transcription/translation as reported by Czajka et al. in DM patients [12].
Second, peripheral leukocyte mtDNA copy number could not precisely reflect mitochondrial activity of the body. The gold standard is to evaluate the mtDNA activity both in muscle and adipose tissue. One proposed explanation why the mtDNA copy number has a correlation with BMI but not with total body fat and visceral fat is that measuring BMI would not only count the fat but also the muscle.
This study has some limitations. First, the cross-sectional design in our study can not determine the causal relationship of our result. The small sample size is also another limitation of our study, therefore further studies with larger sample sizes are needed to confirm our findings. We did not perform fat and muscle biopsy as the gold standard in the evaluation of mitochondrial function. We also did not measure the mitochondrial RNAs expression which was coded by mtDNA copy number, therefore we suggest further studies to be conducted on this subject .
CONCLUSION
Our study shows that peripheral blood mtDNA copy number is associated with body mass index and waist circumference in 25 healthy young adult males (19-24 years) although the causality of our findings cannot be determined. Further studies are needed to understand pathophysiology and causality of our finding.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by Komite Etik Penelitian Kesehatan (Health Research Ethical Committee) of Medical Faculty, Hasanuddin University.
HUMAN AND ANIMAL RIGHTS
No animals were used in this study. This study complied with the Declaration of Helsinki.
CONSENT FOR PUBLICATION
Written informed consent was obtained from each participant.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise. This article is a part of Corresponding Author Dissertation Project.
ACKNOWLEDGEMENTS
The authors are grateful to all Clinical Pathology Residents, Faculty of Medicine, Hasanuddin University, for their help and cooperation in the preparation of samples. We would like to thank the Ministry of Research, Technology, and Higher Education, Republic of Indonesia and Hasanuddin University for aiding funds.




